Opioids and Blockchain; How Technology can help in the fight against a Drug Epidemic
Opioids such as Fentanyl were the cause of 63,000 overdoes deaths in 2017 (CDC Statistics) which is a rise of over 11,300 from the year before. Fentanyl itself is approximately 35 times more powerful then heroin and thus a deadly factor when combined with counterfeit drugs. According to the February 2018 National Association of Boards of Pharmacy report on Internet Drug Outlet Identification Program there are over 11,000 drug sites found to be operating out of compliance with the US federal and state pharmacy laws. Furthermore, they evaluated 100 websites during the second half of 2017 and found all to be operating illegally, offering prescription drugs and controlled substances without a prescription. If this is the case, there is no doubt that the opioids and fentanyl being sold there would be unregulated and most likely counterfeit. The two of these together are a deadly combination which precipitated many of the overdose deaths mentioned earlier.
Many of these compromised drugs can be found online at e-pharmacy sites that aren’t particularly if at all licensed and regulated. We often hear how good Canadian medicine is as well as the cost effectiveness of their pharmacies. This had lead to an uptick in purchasing from Canadian pharmacies online, however according to the Alliance for Safe Online Pharmacies, 74% of “Canadian Online Pharmacies” source their drugs from outside of Canada. From 2010 to 2014, Interpol’s Operation Pangea, with support from nations all over the globe, suspended 57,000 illicit online pharmacies and seized more than 30.3 million units of fake medications. This trend has grown and according to the WHO, 100,000 to 1 million people die from fake modification each year with this being an over 200-billion-dollar market. While fighting drug addiction is very difficult and must be implemented in a multi-faceted manner, the fight against illegal and fake medications reaching the hands of the end user is easier to confront. The beginning of this fight against compromised drugs begins with ensuring the pharmaceutical supply chain.
The supply chain of a drug follows these basic 8 steps; 1. origination of components of drugs (natural, chemical, synthetic etc.) 2. Transportation of drugs to the manufacturer 3. Making of the appropriate lab/factory etc 4. Transportation of the drugs to the wholesale distributor 5. Transportation of the drugs to the repackager 6. Returning of the packaged drugs to the wholesale 7. Transportation of drugs to pharmacy or hospital 8. Distribution of drugs to patient. Throughout this entire system there needs to be trust and conversations between all the different parties for the drug to be original to what the actual patient needs and not faked or switched along the way. There is a way to do this, however we must first see where most of the pharmaceutical drugs in the supply chain come from.
Controlling these steps are several conglomerates who in large part control the pharmaceutical market, and they are;
Within these exports, the following conglomerates control the majority portion of the market;
§ Pfizer (United States)
§ Novartis (Switzerland)
§ Sanofi (France)
§ Roche Holding (Switzerland)
§ Merck & Co (United States)
§ GlaxoSmithKline (United Kingdom)
§ AstraZeneca (United Kingdom)
§ Eli Lilly & Co (United States)
§ Abbott Laboratories (United States)
§ McKesson (United States)
§ AbbVie (United States)
§ Bristol-Myers Squibb (United States)
§ Teva Pharmaceutical (Israel)
§ Takeda Pharmaceutical (Japan)
§ Merck (Germany)
§ Novo Nordisk (Denmark)
§ Cardinal Health (United States)
§ Otsuka Holding (Japan)
§ Astellas Pharma (Japan)
§ AmerisourceBergen (United States)
§ Daiichi Sankyo (Japan)
§ Allergan (United States)
§ Mylan (United States)
§ Actavis (Ireland)
§ Sinopharm Group (China)
§ Valeant Pharmaceuticals (Canada)
Concurrently the 15 countries that exported the highest dollar value worth of drugs and medicines during 2017:
1. Germany: US$52.7 billion (16% of total drugs/medicines exports)
2. Switzerland: $41 billion (12.5%)
3. Belgium: $26.5 billion (8.1%)
4. France: $24.4 billion (7.4%)
5. United Kingdom: $20.4 billion (6.2%)
6. United States: $20.1 billion (6.1%)
7. Italy: $18.3 billion (5.6%)
8. Netherlands: $17.6 billion (5.4%)
9. Ireland: $17.2 billion (5.2%)
10. Denmark: $11.3 billion (3.4%)
11. India: $8.7 billion (2.6%)
12. Spain: $7.7 billion (2.3%)
13. Sweden: $6.2 billion (1.9%)
14. Austria: $5.3 billion (1.6%)
15. Canada: $5.1 billion (1.6%)
By value, the listed 15 countries shipped 85.3% of all exported drugs and medicine for 2017. (information taken from www.worldstopexports.com)
Because we know which companies control the pharmaceutical market and which counties export those drugs, we can look for technology whereby in partnership they could track drugs from creation to consumer thus eliminating in a large part counterfeit drugs. Once these counterfeit drugs are eliminated the opioid black-market crisis can be severely hampered. For we would be able to track the drugs and know what is legitimate and what is not. Fourth generation technology is available to fight the counterfeiting of drugs coming into the country.
In our estimation, Blockchain Technology is an integral factor in the fight against counterfeit of drugs coming into the country. The technology that I am speaking about is in its infancy but holds the prospect of radically changing “the system” to the benefit of the entire health care system. The technology that I am speaking of is called Blockchain. A blockchain is a distributed ledger whereby every ledger holds the same information and all of them fit together one piece at a time. Furthermore, this distributed electronic (digital) ledger writes and publicizes all the public information that is written on it to each of the ledgers, thus ensuring that it is accurate and can’t be lost. The blocks of information can also be put on a private permissioned ledger if so needed. Since the ledger is immutable and can’t be changed all the knowledge within it is known and permanent. This fact alone would be a huge help in fighting counterfeit and dangerous drugs.
With the blockchain, all pharmaceutical drug supply information would be logged within the ledger, updated in real time, distributed upon a network and verifiable at any time. The fact that it is distributed ensures that every step is agreed upon to be true and the drug that is being delivered is original all along its path from one to eight. The fact that the trust is eliminated from the equation due to the technology and its distributed verification ensures the legitimacy of the drug and that the patient is receiving that which they have been prescribed.
In connection with this a universal smart contract could be used in order to track the supply chain and work in conjunction with the blockchain. On one hand the blockchain would track and store all information and the smart contract would control the steps along the process. A smart contract itself. Smart contracts which were first written about in 1994 by Nick Szabo were defined by him as computerized transaction protocols that execute terms of a contract. In the realm of Suppliers, manufacturing, distribution groups and retail groups (this being in line with the drug supply chain) the process would look like the following diagram.
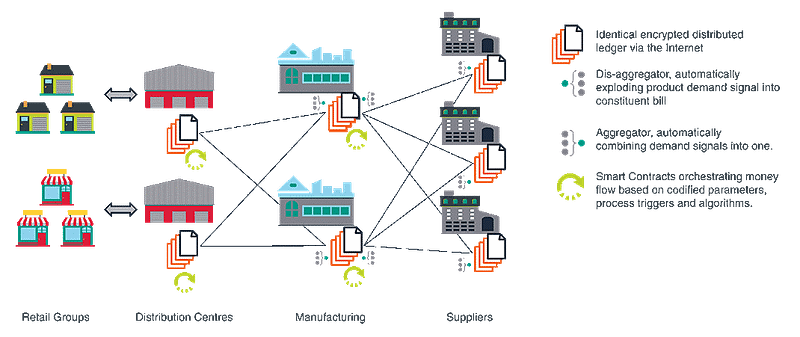
As we can see the ability to electronically track and verify each step along the process would ensure the safety, reliability and authenticity of today’s modern pharmaceutical ecosystem. It is now up to big pharma to ensure this authenticity through the creation of a model industry wide smart contract and blockchain ecosystem. For once this is done we will be better able to trace the authenticity of drugs and fight counterfeiting. Thus ensuring the safety of these drugs an cutting down on overdose’s due to higher then acceptable levels of ingredients such as fentanyl.





Thank you for posting. Good information. I look forward to reading more.